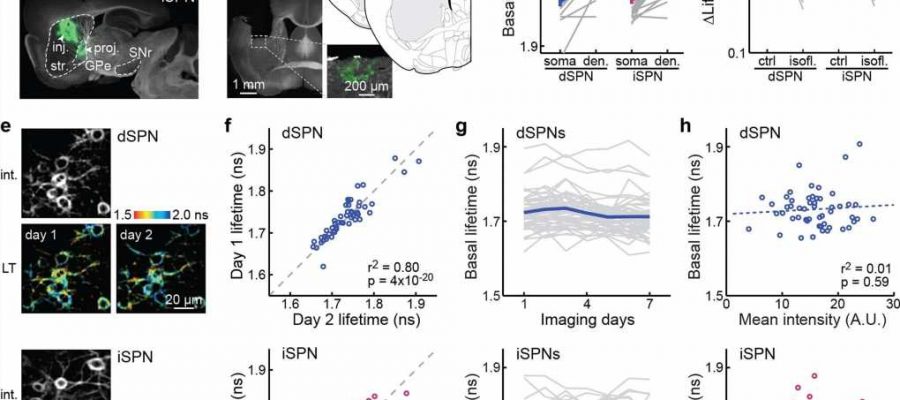
![dSPNs and iSPNs exhibit cell-specific set points of basal PKA activity. <b>a</b>, Example projections from dSPNs and iSPNs, as visualized by Cre-dependent GFP expression in the dorsal striatum of <i>Drd1a-cre</i> (5 mice) and <i>Adora2a-cre</i> mice (2 mice), respectively. GPe: globus pallidus, external segment; SNr: substantia nigra pars reticulata; inj: injection site; proj: projection site. <b>b</b>, Representative post hoc histology section of an animal with GRIN lens implanted and tAKARα expressed in iSPNs, and centers of GRIN lens implantations for dSPNs (blue, 17 mice) and iSPNs (magenta, 15 mice), as mapped onto nearby coronal sections from Franklin & Paxinos (2007). Section positions in millimeters anterior to bregma are indicated. <b>c</b>, Comparison of basal tAKARα lifetimes between dSPN or iSPN somata and their corresponding dendrites. n (neurons/mice) = 16/4 for dSPN and 22/6 for iSPN. Between somata and dendrites, two-tailed paired Student’s t-test, from left to right, p = 1.4x10<sup>−4</sup> and 1.5x10<sup>−4</sup>; dF = 15 and 21; t = 5.1 and 4.6. Between somata of dSPNs and iSPNs, two-tailed unpaired Student’s t-test, p = 0.79; dF = 36; t = 0.26. <b>d</b>, Collective changes of basal PKA activity responding to isoflurane (isofl.) exposure (1.5%). n (neurons/mice) = 52/5 for dSPNs and 71/7 for iSPNs. Two-tailed paired Student’s t-test, from left to right, p = 5.8x10<sup>−25</sup> and 3.5x10<sup>−32</sup>; dF = 51 and 70; t = −19.2, and −21.2. <b>e</b> & <b>f</b>, Representative intensity and corresponding lifetime (LT) images across two days (<b>e</b>) and correlation of basal lifetimes of the same cells (<b>f</b>) for dSPNs and iSPNs. The p values are from the fit. <b>g</b>, Basal lifetimes of the same cells across seven consecutive days. <b>h</b>, Correlation of basal lifetimes with the average fluorescence intensity of the corresponding cells. The p values are from the fit. For panels <b>e</b>–<b>h</b>: n (neurons/fields of view [FOVs]/mice) = 52/12/5 for dSPNs and 86/19/8 for iSPNs. All error bars represent SEM and their centers represent the mean. n.s.: p > 0.05; ***: p ≤ 0.001. Credit: <i>Nature</i> (2022). DOI: 10.1038/s41586-022-05407-4 Scientists identify molecule that could help treat Parkinson's](https://scx1.b-cdn.net/csz/news/800a/2022/scientists-identify-mo.jpg)
Researchers at Oregon Health & Science University have discovered that the neurotransmitter adenosine effectively acts as a brake to dopamine, another well-known neurotransmitter involved in motor control.
Scientists found that adenosine operates in a kind of push-pull dynamic with dopamine in the brain; the discovery published today in the journal Nature.
“There are two neuronal circuits: one that helps promote action and the other that inhibits action,” said senior author Haining Zhong, Ph.D., scientist with the OHSU Vollum Institute. “Dopamine promotes the first circuit to enable movement, and adenosine is the ‘brake’ that promotes the second circuit and brings balance to the system.”
The discovery could immediately suggest new avenues of drug development to treat symptoms of Parkinson’s disease, a movement disorder where the loss of dopamine-producing cells has been widely implicated as a cause.
Scientists have long suspected that dopamine is influenced by an opposing dynamic of neuronal signaling in the striatum—a critical region of the brain that mediates movement along with reward, motivation and learning. The striatum is also the primary brain region affected in Parkinson’s disease by the loss of dopamine-producing cells.
“People for a long time suspected there has to be this push-pull system,” said co-author Tianyi Mao, Ph.D., a scientist at the Vollum who happens to be married to Zhong.
In the new study, researchers for the first time clearly and definitively revealed adenosine as the neurotransmitter that acts in an oppositional sense with dopamine. The study, involving mice, used novel genetically engineered protein probes recently developed in the Zhong and Mao labs. An example of that technology was highlighted last month in a study published in the journal Nature Methods.
Notably, adenosine is also well known as the receptor that caffeine acts upon.
“Coffee acts in our brain through the same receptors,” Mao said. “Drinking coffee lifts the brake imposed by adenosine.”
More information:
Haining Zhong, Locomotion activates PKA through dopamine and adenosine in striatal neurons, Nature (2022). DOI: 10.1038/s41586-022-05407-4
Crystian I. Massengill et al, Sensitive genetically encoded sensors for population and subcellular imaging of cAMP in vivo, Nature Methods (2022). DOI: 10.1038/s41592-022-01646-5
Journal information:
Nature Methods
,
Nature
Source: Read Full Article