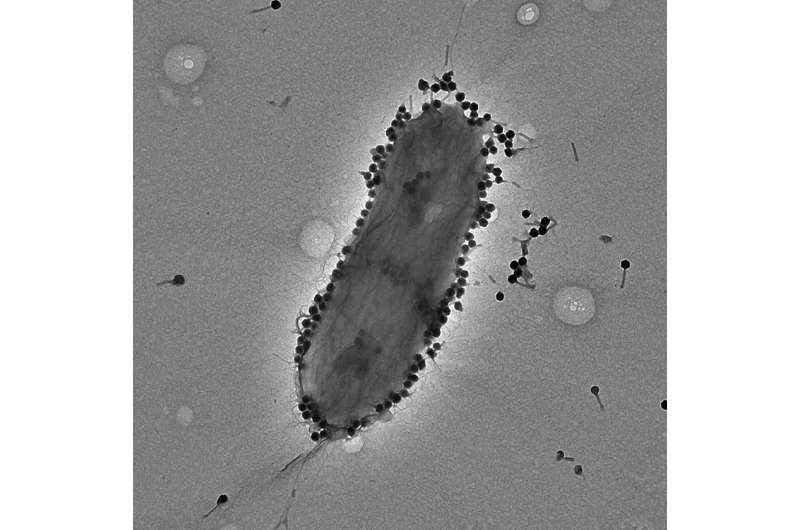
A new international study conducted by the Israeli Phage Therapy Center (IPTC) led by Prof. Ran Nir-Paz at Hadassah Hebrew University Medical Center and Prof. Ronen Hazan of the Faculty of Dental Medicine at The Hebrew University of Jerusalem, using phage PASA16 on compassionate basis to treat tough Pseudomonas aeruginosa infections, has shown promising results with an 86.6% success rate.
This research involving 16 patients with persistent infections represents the largest of its kind and brings encouraging findings. The study’s success demonstrates the potential effectiveness of PASA16 phage therapy in tackling challenging Pseudomonas aeruginosa infections, paving the way for future clinical trials and encouraging further exploration of phage therapy as an alternative and auxiliary approach against antibiotic-resistant infections. The paper is published in the journal Med.
Phage therapy, the use of specific anti-bacterial viruses against infections, has garnered attention as a critical addition to conventional antibiotics, though clinical trials in this area have been limited. However, recent compassionate phage therapy cases have shown potential, albeit with scarce evidence on treating difficult infections.
This study sheds light on the potential role of phages in combination with antibiotics in combating the hard-to-treat pathogen P. aeruginosa infections that were unresponsive to conventional treatments.
Prior to treatment, all P. aeruginosa isolates from patients were tested and treatment was personalized provided to those who were found to be sensitive to the PASA16 phage. During the PASA16 phage treatment, only minor side effects were observed, which were manageable. Remarkably, 13 out of 15 patients with available data achieved a favorable clinical outcome, resulting in an impressive 86.6% success rate.
This highlights the potential of combining PASA16 phage with antibiotics as a promising approach for patients with previously unsuccessful treatments. The duration of treatment spanned from eight days to six weeks (majority of around two weeks), with one- to twice-daily regimens, offering a time-efficient option.
In conclusion, this compassionate use case series demonstrates the potential effectiveness of PASA16 phage therapy in tackling tough Pseudomonas aeruginosa infections. By outlining potential clinical protocols, this study paves the way for future trials. The success observed encourages further research and exploration of phage therapy as an alternative and complementary approach to combat antibiotic-resistant infections.
Prof. Ran Nir-Paz, co- lead researcher of the study at IPTC of Hadassah Hebrew University Medical Center, expressed excitement about the findings, stating, “We are elated by the promising results of our study using phage PASA16 to treat tough Pseudomonas aeruginosa infections. This groundbreaking research offers hope for patients with persistent infections and highlights the potential of phage therapy as a valuable alternative to conventional antibiotics in combating antibiotic-resistant pathogens.”
Prof. Ronen Hazan the co-lead researcher of the IPTC, also the head of the Bio-research institute of the Faculty of Dental Medicine at The Hebrew University of Jerusalem said, “We are encouraged by the findings. The study’s 86.6% success rate offers hope for patients with persistent infections and underscores the potential of phage therapy as an alternative to combat antibiotic-resistant pathogens.”
P. aeruginosa is a type of bacteria commonly found in the environment, such as soil, water, and plants, and as part of bacteria residing in humans. It is both a pathogen and opportunistic bacteria, causing infections in individuals with weakened immune systems or underlying health conditions. Known for its potentially complicated and life-threatening infections, P. aeruginosa poses challenges in health care settings.
These infections can range from mild to severe, affecting various body parts, including the lungs, urinary tract, skin, and wounds. It is a common cause of hospital-acquired infections, particularly in immunocompromised patients or those using mechanical ventilation or invasive devices. The bacteria’s ability to form protective biofilms hinders treatment, sometimes necessitating the combination of antibiotics and alternative therapies like phage therapy. Strict infection control measures in health care facilities are essential to combat its persistence.
The study compiled comprehensive clinical data on 15 out of 16 patients treated with phage PASA16, assessing its efficacy in treating the infections. The phage, provided pro bono by the American phage company “Adaptive Phage Therapeutics,” was administered through various methods, including intravenous, local application to the infection site, and topical use. The data mainly covered patients with osteoarticular and foreign-device-associated infections.
As the fight against antibiotic resistance continues, this compassionate use case series represents a significant step forward in exploring phage therapy as a potential solution for patients with tough P. aeruginosa infections.
Phage therapy has re-emerged as a potential solution for antimicrobial-resistant and non-resolving infections, as phages specifically target other microorganisms. While compassionate use cases for phage therapy have been implemented, clinical trials remain limited.
This study reports the largest case series of consecutive patients with severe P. aeruginosa infections who received compassionate use treatment using a single specific phage, PASA16. All 16 patients were treated after demonstrating the susceptibility of their infective agent to both the phage alone and in combination with antibiotics. The study findings demonstrate a favorable outcome in over 80% of treated patients, with minimal side effects. Based on these findings, further compassionate use cases and the development of clinical trial protocols should follow.
More information:
Ran Nir-Paz, Refractory Pseudomonas aeruginosa infections treated with Phage PASA16: a compassionate use case series, Med (2023). DOI: 10.1016/j.medj.2023.07.002. www.cell.com/med/fulltext/S2666-6340(23)00225-8
Journal information:
Med
Source: Read Full Article
