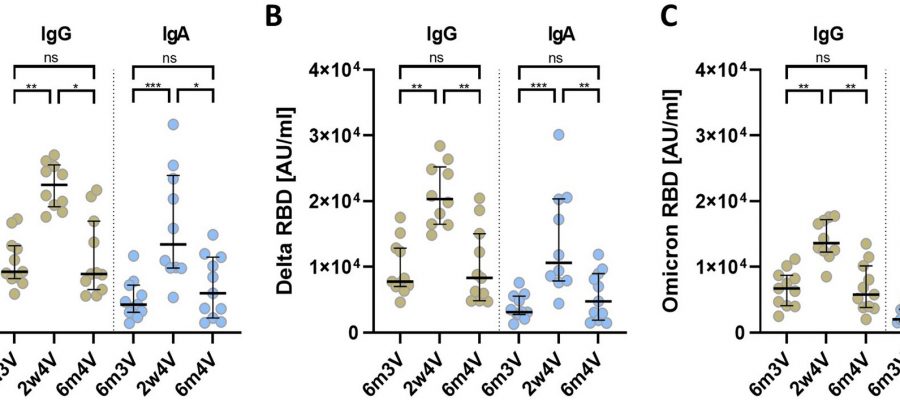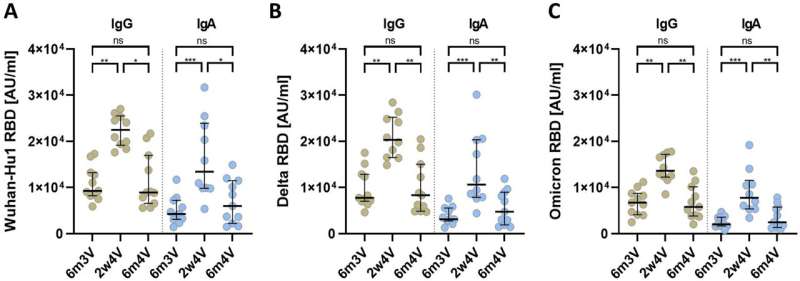
Booster vaccinations (third and fourth vaccinations) of COVID-19 mRNA vaccines that are not adapted to omicron variants do lead to higher blood levels of neutralizing antibodies against the omicron subvariants. However, these antibody titers drop significantly six months after the third or fourth vaccination. The fourth vaccination also had no effect on the breadth of the immune response against different virus variants.
Even with primary immunization with Comirnaty, only small amounts of neutralizing antibodies against omicron were present. These are the results of a clinical study conducted by the Paul-Ehrlich-Institut in vaccinated health care professionals. The research is published in the journal Scientific Reports.
Booster vaccination to strengthen immunological memory
Booster shots are recommended in most age groups to protect against COVID-19. Boosters are administered for several reasons: antibody titers rise rapidly within the first two weeks after a booster vaccination and offer the best protection against COVID-19. The antibody titer then drops to a baseline level after the first few months. This baseline level is built up or refreshed by the immunological memory, resulting in an increase in memory B cells and long-lived plasma cells, which in turn form antibodies.
Both cell types are important pillars of the antibody-based humoral immune response. Booster vaccination also stimulates a broader humoral immune response, which is produced by differentiation processes of the antibody genes (somatic hypermutation) and antibody affinity maturation.
SARS-CoV-2 omicron variants necessitate vaccine adaptations
Omicron subvariants of the SARS-CoV-2 coronavirus have dominated since the beginning of 2022. The omicron subvariants have an enormous escape potential due to the destruction or deletion of different areas of the virus that are important for the immune response (epitopes). As a result, the corresponding virus variants evade the immune response.
This escape potential also explains why the number of breakthrough infections in vaccinated individuals has increased significantly since the occurrence of the omicron subvariants: Due to the numerous mutations in the spike protein, many of the antibodies induced by vaccination and/or infection cannot bind to the mutated spike protein and thus do not develop their neutralizing potential.
Clinical study shows non-omicron adapted COVID-19 vaccines don’t provide continued high antibody levels against omicron variants
A research team at the Paul-Ehrlich-Institut led by Professor Eberhard Hildt, head of the Virology Division, investigated the influence of the original mRNA vaccines Comirnaty (BioNTech) and Spikevax (Moderna), which had not yet been adapted to the omicron variants, on the levels of protection against omicron variants offered by neutralizing antibodies in a clinical study.
After primary immunization (Comirnaty), the subjects had low blood levels (titers) of neutralizing antibodies to omicron. Booster vaccinations (3rd and 4th vaccinations) with the monovalent mRNA vaccine Spikevax, also not yet adapted to omicron variants at the time, did lead to higher titers of neutralizing antibodies against the omicron variants.
However, the antibody titers also dropped six months after the fourth vaccination (2nd booster vaccination)—just as they did after the third vaccination. The fourth vaccination also had no effect on the breadth of the antibody-based immune response, which gives an indication of the extent to which there is protection against infection with various virus variants.
The results of this study also point to the importance of the COVID-19 booster vaccines adapted to omicron subvariants.
More information:
Sascha Hein et al, The fourth vaccination with a non-SARS-CoV-2 variant adapted vaccine fails to increase the breadth of the humoral immune response, Scientific Reports (2023). DOI: 10.1038/s41598-023-38077-x
Journal information:
Scientific Reports
Source: Read Full Article