SIMBRINZA®
brinzolamide 1% and brimonidine tartrate 0.2%
CONSUMER MEDICINE INFORMATION
What is in this leaflet
Please read this leaflet carefully before you use Simbrinza Eye Drops.
This leaflet answers some common questions about Simbrinza Eye Drops. It does not contain all of the available information. It does not take the place of talking to your doctor or pharmacist.
The information in this leaflet was last updated on the date listed on the final page. More recent information on the medicine may be available.
You should ensure that you speak to your pharmacist or doctor to obtain the most up to date information on the medicine.
You can also download the most up to date leaflet in Australia from www.novartis.com.au and www.medsafe.govt.nz in New Zealand.
The updates may contain important information about the medicine and its use of which you should be aware.
All medicines have risks and benefits. Your doctor has weighed the expected benefits of you using Simbrinza Eye Drops against the risks this medicine could have for you.
The information in this leaflet applies to Simbrinza only. This information does not apply to similar products, even if they contain the same ingredients.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine.
You may need to read it again.
What SIMBRINZA is used for
Simbrinza is used to lower pressure in the eye and to treat open-angle glaucoma or ocular hypertension.
Glaucoma is a condition in which the pressure of fluid in the eye may be high. However, some people with glaucoma may have normal eye pressure.
Glaucoma is usually caused by a build up of the fluid which flows through the eye. This build up occurs because the fluid drains out of your eye more slowly than it is being pumped in. Since new fluid continues to enter the eye, joining the fluid already there, the pressure continues to rise. This raised pressure may damage the back of the eye resulting in gradual loss of sight. Damage can progress so slowly that the person is not aware of this gradual loss of sight. Sometimes even normal eye pressure is associated with damage to the back of the eye.
There are usually no symptoms of glaucoma. The only way of knowing that you have glaucoma is to have your eye pressure, optic nerve and visual field checked by an eye specialist. If glaucoma is not treated, it can lead to serious problems, including total blindness. In fact, untreated glaucoma is one of the most common causes of blindness.
Simbrinza is not suitable for use in acute angle-closure glaucoma.
Simbrinza combines two ingredients, brinzolamide and brimonidine tartrate. Brinzolamide is a carbonic anhydrase inhibitor and brimonidine tartrate is an alpha-2 adrenergic agonist. Both brinzolamide and brimonidine tartrate work by reducing the production of fluid within the eye.
For more information about glaucoma in Australia, contact Glaucoma Australia (telephone 1800 500 880). In New Zealand contact Glaucoma New Zealand on 09 373 8779 or free phone on 0800 GLAUCOMA or 0800 452 826.
Your doctor may have prescribed Simbrinza for another reason. Ask your doctor if you have any questions about why Simbrinza has been prescribed for you.
This medicine is available only with a doctor’s prescription.
There is no evidence that Simbrinza is addictive.
Use in Children
Simbrinza Eye Drops are not recommended in children and young adults aged between 2 to 17 years as the safety and efficacy in children has not been established.
Simbrinza Eye Drops should not be used in children under 2 year of age.
Before you use SIMBRINZA
When you must not use it
Do not use Simbrinza if you have an allergy to:
brinzolamide
brimonidine tartrate
any other carbonic anhydrase inhibitor or alpha-2 adrenergic agonist
any of the ingredients listed at the end of this leaflet.
If you are unsure if you are taking or using any of these medicines ask your doctor.
Some of the symptoms of an allergic reaction may include:
shortness of breath
wheezing or difficulty breathing
swelling of the face, lips, tongue or other parts of the body
rash, itching or hives on the skin.
Do not use Simbrinza if:
you have severe kidney problems
you have too much acidity in your blood (a condition called hyperchloraemic acidosis).
Do not use Simbrinza if:
you are taking any antidepressant medicines.
These include medicines called monoamine oxidase (MAO) inhibitors or other types of antidepressants.
Do not use this medicine after the expiry date printed on the pack or bottle, if the seal around the pack is broken or shows signs of tampering.
If it has expired or is damaged, return it to your pharmacist for disposal.
If you use this medicine after the expiry date has passed, it may not work.
If you are not sure whether you should start using Simbrinza, talk to your doctor.
Before you start to use it
Tell your doctor if you have had an allergy to any other medicines or any other substances, such as foods, preservatives or dyes.
Tell your doctor if you have an allergy to sulphonamide medicines.
Brinzolamide, one of the active ingredients in Simbrinza, is a sulfur-containing medicine. If you are allergic to sulfur medicines, such as some antibiotics, you may be allergic to Simbrinza Eye Drops. Check with your doctor or pharmacist if you are not sure whether you are allergic to sulphonamides.
Tell your doctor if you have or have had any medical conditions, in particular the following:
heart conditions.
These include coronary heart disease (symptoms can include chest pain or tightness, breathlessness or choking), heart failure, high or low blood pressure.
circulation conditions.
These include Raynaud’s disease, thromboangiitis obliterans (Buerger disease) where there is inflammation and clotting of the small and medium arteries and veins of the hands and feet and poor circulation of blood in the brain.
kidney problems
liver problems
compromised cornea and/or corneal disease
depression.
Tell your doctor if you are pregnant or plan to become pregnant.
Your doctor will discuss the possible risks and benefits of using Simbrinza during pregnancy.
Tell your doctor if you are breast-feeding.
Your doctor will discuss the possible risks and benefits of using Simbrinza when you are breast-feeding.
If you have not told your doctor about any of the above, tell him/her before you use Simbrinza.
Taking or using other medicines
Tell your doctor if you are taking or using any other medicines, including other eye drops or ointments, and any other medicines that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Simbrinza may interfere with each other or add to the effects of Simbrinza. These include:
medicines to lower blood pressure
digoxin, a medicine used to treat heart failure
other medicines or eye drops for glaucoma
carbonic anhydrase inhibitors such as acetazolamide, dorzolamide
antiviral or antibiotic medicines
chlorpromazine, methylphenidate, reserpine
isoprenaline, prazosin
salicylates such as aspirin
monoamine oxidase (MAO) inhibitors, or other antidepressants including amitriptyline, nortriptyline, clomipramine and mianserin
CNS depressants such as alcohol, barbiturates, opiates, sedatives or anaesthetics.
These medicines may be affected by Simbrinza, or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor or pharmacist has more information on medicines to be careful with or avoid while using Simbrinza.
How to use SIMBRINZA
Carefully follow all directions given to you by your doctor and pharmacist.
They may differ from the information contained in this leaflet.
If you are being changed from one medicine to another, follow your doctor’s instructions carefully as to when to stop the medicine and when to start the new eye drops.
If you do not understand the instructions on the carton, ask your doctor or pharmacist for help.
How much to use
Use Simbrinza only when prescribed by your doctor.
The usual dose of Simbrinza is one drop in the affected eye(s) twice daily. Usually, this means one drop in the affected eye(s) first thing in the morning, and one drop in the evening.
Your doctor will tell you how many drops you need to use each day.
These dosing instructions will be printed on the label your pharmacist adds.
How to use it
It is important to use Simbrinza exactly as your doctor or pharmacist has told you. If you use the drops less often than prescribed, they may not work as well and the eye problem may not improve. Using the drops more often than prescribed may not improve the eye problem any faster and may cause increased side effects.
If you are wearing soft contact lenses, remove them before putting the drops in your eye.
Follow these steps to use the eye drops:
1.Wash your hands thoroughly with soap and water.
2.Immediately before using a bottle for the first time, break the safety seal around the neck area and throw the loose plastic ring away.
4.Remove the cap from the bottle.
5.Hold the bottle upside down in one hand between your thumb and middle finger (see Diagram 1).
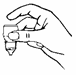
Diagram 1
6.While tilting your head back, gently pull down the lower eyelid of your eye to form a pouch/pocket.
7.Place the tip of the bottle close to your eye. Do not let it touch your eye. Do not squeeze the bottle.
8.Release one drop into the pouch/pocket formed between your eye and eyelid by gently tapping or pressing the base of the bottle with your forefinger (see Diagrams 2 and 3).
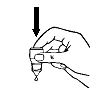
Diagram 2
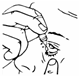
Diagram 3
9.Close your eye. Do not blink or rub your eye.
10.While your eye is closed, place your index finger against the inside corner of your eye and press against your nose for about two minutes. This will help to stop the medicine from draining through the tear duct to the nose and throat, from where it can be absorbed into other parts of your body. This will also reduce the unpleasant taste sensation that some people experience when using these drops.
11.If necessary, repeat the above steps for the other eye.
12.Your eyelids can only hold less than one drop at a time, so it is normal for a small amount of the eye drop to spill onto your cheek. You should wipe away any spillage with a tissue.
13.Replace the cap on the bottle, closing it tightly.
14.Wash your hands again with soap and water to remove any residue.
You may feel a slight burning sensation in the eye shortly after using the eye drops. If this persists, or is very uncomfortable, contact your doctor or pharmacist.
Be careful not to touch the dropper tip against your eye, eyelid or anything else.
This will help prevent the drops becoming dirty or contaminated.
If you have trouble knowing whether you have placed your drops correctly, you may want to store them in the fridge. Some people find it easier to feel the drops in the eye if they are cold.
After using Simbrinza, wait at least 5 minutes before putting any other eye drops in your eye(s).
Wait 15 minutes before replacing your contact lenses.
When to use it
Use Simbrinza every day, at about the same times each day, unless your doctor tells you otherwise. Using your eye drops at the same times each day will have the best effect on your eye pressure. It will also help you remember when to use the eye drops.
How long to use it
Simbrinza helps control your condition but does not cure it. Simbrinza must be used every day. Continue using Simbrinza for as long as your doctor prescribes.
If you are unsure about when or how to stop using Simbrinza, you should talk to your doctor or pharmacist.
If you forget to use it
If it is almost time for your next dose, skip the dose you missed and use your next dose when you are meant to.
Otherwise, use the drops as soon as you remember, and then go back to using them as you would normally.
Do not use double the amount to make up for the dose that you missed.
Using multiple doses may cause unwanted side effects.
If you are not sure whether to skip the dose, talk to your doctor or pharmacist.
If you have trouble remembering to use your eye drops, ask your pharmacist for some hints.
If you use too much (overdose)
If you accidentally put several drops in your eye(s), immediately rinse your eye(s) with warm water.
If you think that you or anyone else may have swallowed any or all of the contents of a bottle of Simbrinza, immediately telephone your doctor or the Poisons Information Centre on 13 1126 in Australia or the National Poisons Centre in New Zealand on 0800 POISON or 0800 764 for advice. Alternatively go to Accident and Emergency at your nearest hospital. Do this even if there are no signs of discomfort or poisoning.
While you are using SIMBRINZA
Things you must do
Have your eye pressure checked when your eye specialist says, to make sure Simbrinza is working.
If you are about to start taking or using any new medicine, tell your doctor and pharmacist that you are using Simbrinza.
Tell all the doctors, dentists and pharmacists who are treating you that you are using Simbrinza.
Tell your doctor if, for any reason, you have not used Simbrinza exactly as prescribed. Otherwise your doctor may think that the eye drops were not effective and change the treatment unnecessarily.
If you become pregnant or decide to breast-feed while using Simbrinza, tell your doctor immediately.
Things you must not do
Do not use Simbrinza to treat other complaints unless your doctor tells you to.
Do not give Simbrinza to anyone else, even if they have the same condition as you.
Do not stop using Simbrinza without first talking to your doctor. If you stop using your eye drops, your eye pressure may rise again and damage to your eye may occur.
Do not put the Simbrinza eye drops into your eye(s) while you are wearing soft contact lenses.
The preservative in Simbrinza (benzalkonium chloride) may be deposited in soft contact lenses. You can put your soft contact lenses back into your eyes 15 minutes after you have used Simbrinza.
Things to be careful of
Be careful driving, operating machinery or performing tasks requiring mental alertness and/or physical coordination until you know how Simbrinza affects you and your vision. As with any eye medicine, temporary blurred vision or other visual disturbances may affect the ability to drive or use machinery in some people. If blurred vision occurs when you use your drops, wait until your vision is clear before driving or operating machinery.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using Simbrinza.
Simbrinza helps most people with high eye pressure and glaucoma, but it may have unwanted side effects in a few people.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Do not be alarmed by the following list of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following effects in the eye(s) and the eye area, and they worry you:
blurred vision
eye allergy
eye discomfort
eye pain
eye redness
eye surface inflammation
feeling of having something in the eye.
These are the more common side effects of your medicine. They are usually mild and short-lived.
Additional side effects that are noticed less often include:
eye surface inflammation with surface damage
sensitivity to bright light
excessive tearing
dry eye
eye discharge
redness of the eyelids
tired eyes
inflammation of the eyelids
deposits on the eye surface
eye and eyelid swelling.
Occasionally some people notice unwanted effects in the rest of their body as a result of using Simbrinza. These effects may include:
difficulty sleeping
drowsiness
bad taste in the mouth
headache
dizziness
decreased blood pressure
dry throat
stuffy nose
dry nose
postnasal drip
dry mouth
indigestion
abdominal pain
abnormal sensation in mouth
skin inflammation, redness or itching
medication residue.
If any of the following happen, stop using Simbrinza and tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
skin rash
swelling of the face, hands or feet
wheezing, difficulty in breathing
shortness of breath (dyspnoea, heart failure)
severe and sudden onset of pinkish, itchy swellings on the skin, also called hives or nettle rash
These hypersensitivity reactions can be very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Other side effects not listed above may also occur in some people.
After using SIMBRINZA
Storage
Store Simbrinza in a cool dry place where the temperature stays below 25°C. It is not necessary to store Simbrinza in the refrigerator, but it is acceptable if you prefer to instil cold drops.
Do not store Simbrinza or any other medicine in the car, in the bathroom or in other warm, damp places.
Heat and dampness can destroy some medicines.
Do not leave the top off the bottle for any length of time, to avoid contaminating the eye drops.
Keep it where children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
Write the date on the bottle when you open the eye drops and throw out any remaining solution after four weeks.
Eye drops contain a preservative which helps prevent germs growing in the solution for the first four weeks after opening the bottle. After this time there is a greater risk that the drops may become contaminated and cause an eye infection. A new bottle should then be used.
If your doctor tells you to stop using Simbrinza or they have passed their expiry date, ask your pharmacist what to do with any remaining solution.
Product description
What it looks like
Simbrinza is a liquid (white to off-white uniform suspension) supplied in a pack containing 5 mL in an 8 mL dropper bottle, or 2.5 mL in a 4 mL dropper bottle, (DROP-TAINER®) with screw cap.
Tamper evidence is provided with a safety seal around the neck area of the bottle.
Ingredients
Simbrinza contains the active ingredient(s):
brinzolamide 10 mg/mL
brimonidine tartrate 2 mg/mL.
Simbrinza also contains the inactive ingredients:
benzalkonium chloride (used as a preservative)
propylene glycol
carbomer 974P
boric acid
mannitol
sodium chloride
tyloxapol
purified water
hydrochloric acid and/or sodium hydroxide are added to adjust pH.
Allergens:
May contain benzoates, sulfites and hydroxybenzoates.
Supplier
Simbrinza is supplied in Australia by:
Novartis Pharmaceuticals Australia Pty Limited
ABN 18 004 244 160
54 Waterloo Road
Macquarie Park NSW 2113
Telephone No. 1800 671 203
www.novartis.com.au
Simbrinza is supplied in New Zealand by
Novartis New Zealand Limited
PO Box 99102
Newmarket
Auckland 1149
New Zealand.
Free Phone: 0800 354 335.
Date of preparation
This leaflet was prepared in October 2020.
Australian Register Number
AUST R 219839
© Novartis Pharmaceuticals Australia Pty Limited 2020
® Registered Trademark
Internal document code:
Sib271020c based on PI sib271020i