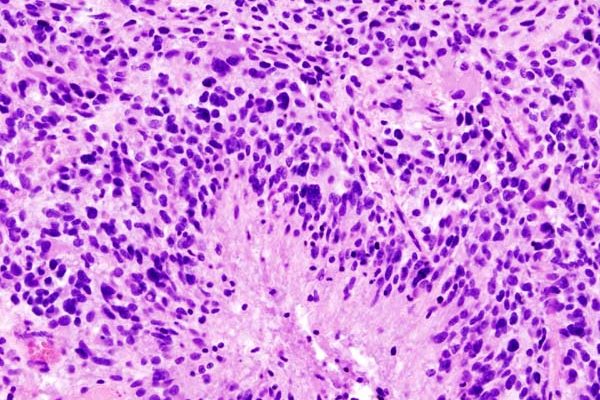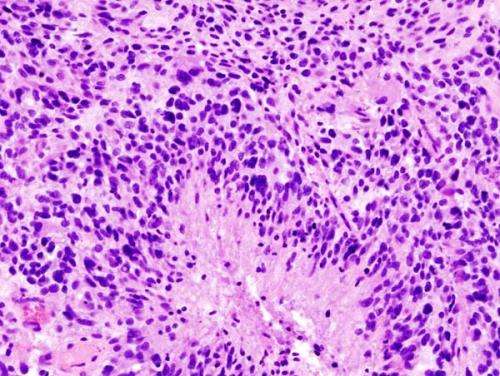
The median length of survival after diagnosis of glioblastoma is 14 months, but some of these brain tumors are more aggressive and resistant to treatment than others, and a new study from Sylvester Comprehensive Cancer Center at the University of Miami Miller School of Medicine suggests reactivation of an ancient retrovirus may be at least partly to blame.
“Our lab found that an evolutionary dormant retrovirus from 6 million years ago, HML-2, a subtype of HERV-K, contributes to brain tumor formation. We demonstrated for the first time that this virus, when reactivated, plays a role in defining the stem-cell state of high-grade gliomas, promoting an aggressive form of cancer,” said neurosurgeon Dr. Ashish Shah, principal investigator at Sylvester’s Brain Tumor Initiative (BTI), and first author of a paper published in the Journal of Clinical Investigation.
Cancer stem cells, a subpopulation of cells in cancers, drive tumor initiation and development and influence how aggressive and resistant to treatment a cancer will be.
This study, conducted by researchers from Sylvester, Georgetown University and the National Institutes of Health, found that HML-2 altered stem cell programming by activating a gene-regulating protein called OCT4. HML-2 was previously implicated in the genesis and development of other cancers, but this is believed to be the first study showing the virus’ effects in gliomas and describing the molecular and cellular mechanisms involved.
“Our results suggest that HML-2 fundamentally contributes to the glioblastoma stem-cell niche, the microenvironment that supports stem cells and determines their fate,” said Dr. Avindra Nath, clinical director of the Division of Intramural Research at the National Institute of Neurological Disorders and Stroke at the National Institutes of Health.
“We conducted a comprehensive translational investigation of HML-2 expression in glioblastoma and its role in maintaining the cancer stem cell phenotype,” said Nath, the article’s senior author. Results were based on analyses of patient-derived glioblastoma cells and mouse model studies.
The team’s findings give researchers targets for developing therapies; and in their work, an anti-retroviral drug significantly reduced HML-2 activity and reduced tumor stem-cell markers, said Shah, director of clinical trials and translational research and principal investigator in the Section of Virology and Immunotherapy at Sylvester Comprehensive Cancer Center’s Brain Tumor Initiative (BTI).
“Targeting the glioblastoma stem cell niche is an attractive option to prevent alterations in stem cells and reduce tumor recurrence and treatment resistance,” he said.
HML-2, a subtype of HERV-K, is one of many human endogenous retroviruses (HERVs), ancestral relics of retroviral infections that occurred throughout history and led to the integration of viral sequences into the human genome. HERVs, making up about 8% of the human genome, are usually silent—unable to cause infection—but scientists have recently discovered that some can be reactivated in certain cancers.
More information:
Ashish H. Shah et al, Human endogenous retrovirus K contributes to a stem cell niche in glioblastoma, Journal of Clinical Investigation (2023). DOI: 10.1172/JCI167929
Journal information:
Journal of Clinical Investigation
Source: Read Full Article